This article summarizes the different post-printing processes to analyze the printed construct’s structure, morphology and functionality. The cover picture of this article shows an example of 3D Bioprinting process steps (source: Regenerative Medicine Frontiers Journal article: Regen Med Front. 2019;1:e190004).

The three basic steps in the 3D bioprinting process include:
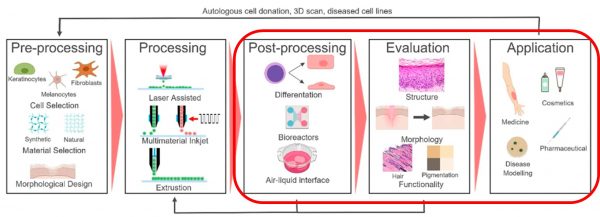
- Pre-printing/pre-processing. This includes the selection of cells and bioink materials, mixing of the ingredients, and creating the 3D design. (This step was covered in the blog post no 2).
- Bioprinting using one or more of the available printing technologies to bioprint a tissue construct and crosslinking to maintain a stable structure. (Part of this step was covered in blog posts no. 3 & 4, and the crosslinking was covered in post no. 5).
- Post-printing. This phase includes the cultivation of the tissue structure in a bioreactor. The printed construct is then mechanically tested and evaluated. Finally, the construct can be used for the intended application. (These steps will be covered in this blog article).
After bioprinting of tissue constructs, more processing is usually required to differentiate the stem cells and maturate the tissue by growing it in a bioreactor or at an air-liquid interface. As cell-cell interactions are highly influenced by post-processing conditions, wide variations of stimuli can be exerted during post-bioprinting processes to modulate the differentiation process.
One major reason for the failure to generate viable bioprinted tissue constructs with clinically relevant thickness in vitro is the lack of adequate oxygen and nutrient supply due to the absence of an internal vascular network. Sufficient oxygen transfer cannot be readily achieved in conventional static tissue culture methods; instead, perfusion in bioreactors is needed. The bioreactor vessel with a pump system assists the cell culture media to flow through the tissue construct to enhance fluid transport. Since external stimuli can also induce differentiation of stem cells into specific cell types, it is essential to exert physiologically-mimetic mechanical and biochemical stimuli on tissue constructs in a bioreactor. This way, the cells can mature into relevant tissues.
After maturation, the tissue construct also needs to be evaluated before application. This stage may include analysis of structure, morphology and function. Optical image analysis can be performed to examine the shape fidelity/quality of the constructs post-printing. The print fidelity is characterized by comparing the experimental, printed dimensions to the theoretical ones.
For many tissues, particularly for load-bearing tissues such as bone and cartilage, the mechanical properties of the printed construct are essential. Hence, a compressive mechanical analysis is performed to evaluate the mechanical stability and compressive modulus of the 3D bioprinted construct. A classic method to study the ability of an object to withstand deformation is to determine the elastic moduli from the stress versus strain curve in compression.
Swelling and degradation kinetics also need to be tuned to match the application. Once the bioprinted construct is placed into either an implanted site or in cell culture media, swelling of the structure occurs. Swelling can influence post-printing mechanics: an increase in fluid increases the distance between crosslinks and decreases crosslink density and thus mechanical properties. But swelling can also be beneficial, as it allows for diffusion of any entrapped therapeutics and cellular waste products. Synthetic bioinks especially must be designed to degrade within an appropriate time scale for the intended application. To fully recapitulate native tissue, degradation profiles are a crucial feature of developed bioinks that must be thoroughly characterized.

In addition to structural and mechanical analysis, it is important to evaluate the effect of shear forces and degradation byproducts on the cells. This is done through various cellular cytotoxicity/viability assays. Concurrent with cellular viability, cell functions such as adhesion, proliferation, and/or differentiation should also be monitored. To understand how cells behave in the printed construct, one can quantify the production of nascent ECM components, such as proteins (collagen, elastin, and fibronectin) and glycosaminoglycans (GAGs) (heparan sulfate, chondroitin sulfate, and so on). In the case of bioprinting, it becomes essential for cells to produce ECM to facilitate further proliferation within the printed construct.
Finally, after all the necessary evaluative steps and possible iterations of all three steps – processing, post-processing, and evaluation – the printed tissue construct can be used for its intended application, such as in vitro disease modelling, cosmetic testing, pharmaceutical testing, or even as a treatment for a particular injury in the human body.
This article is based on the following review articles:
Datta P, Barui A, Wu Y, Ozbolat V, Moncal K, Ozbolat I, Essential steps in bioprinting: From pre- to post-bioprinting, Biotechnology Advances, 2018 36(5): 1481-1504. doi: 10.1016/j.biotechadv.2018.06.003.
Deo KA, Singh KA, Peak CW, Alge DL, Gaharwar AK. Bioprinting 101: Design, Fabrication, and Evaluation of Cell-Laden 3D Bioprinted Scaffolds. Tissue Eng Part A., 2020 Mar;26(5-6):318-338. doi: 10.1089/ten.TEA.2019.0298.